 LL37-dsRNA binding to TLR3
LL37-dsRNA binding to TLR3
Crystallinity of Double-Stranded RNA-Antimicrobial Peptide Complexes Modulates Toll-Like Receptor 3-Mediated Inflammation
Abstract
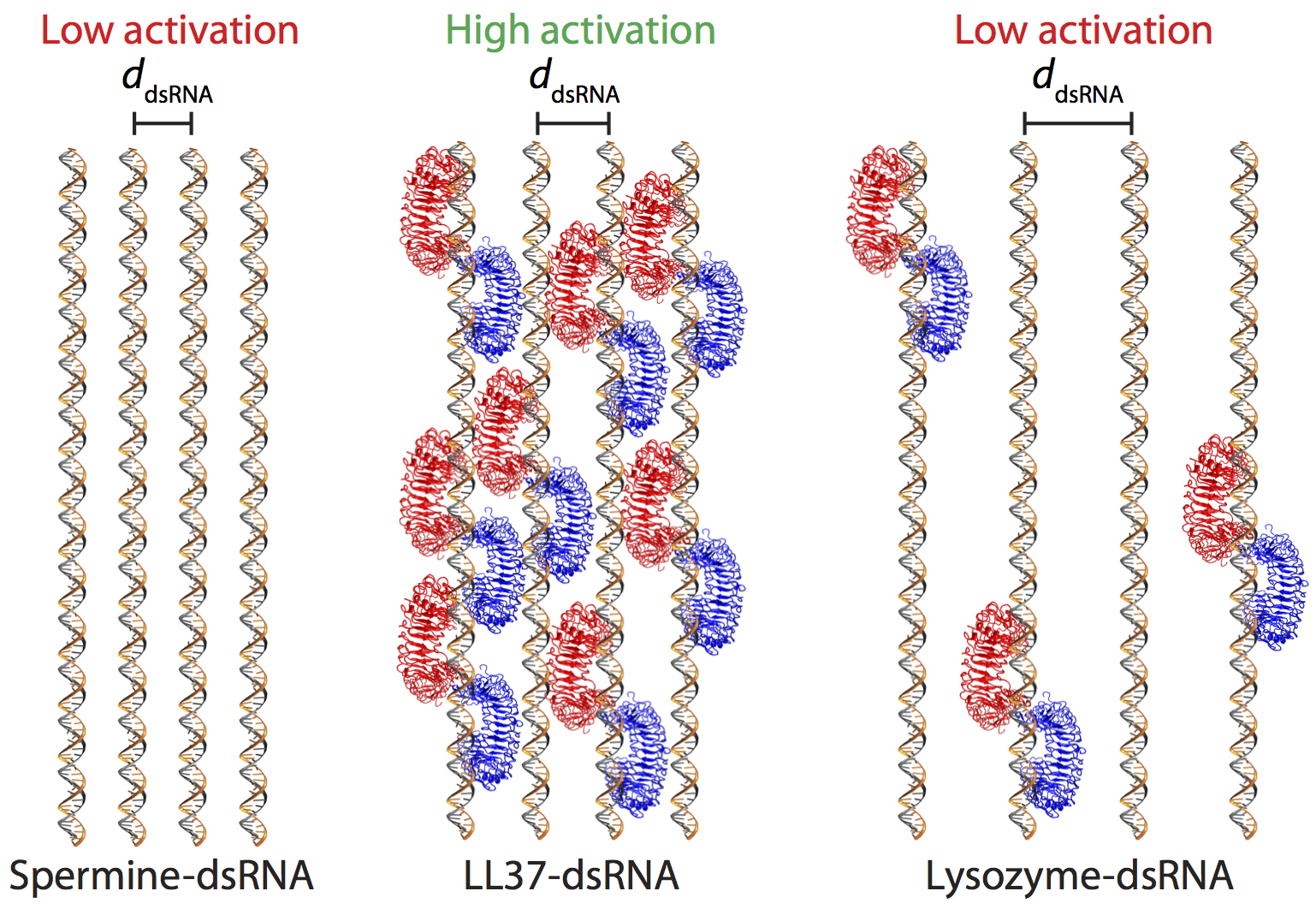
Double-stranded RNA (dsRNA) induces production of pro-inflammatory cytokines in normal human epidermal keratinocytes (NHEK) by specific binding to endosomal Toll-like receptor-3 (TLR3). Recently, it has been shown that hyperactivation of TLR3 in psoriatic keratinocytes by dsRNA can occur in the presence of human antimicrobial peptide (AMP) LL37. Here, we combine synchrotron X-ray scattering, microscopy, computer simulations, and measurements of NHEK cytokine production to elucidate a previously unanticipated form of specific molecular pattern recognition. LL37 and similar α-helical AMPs can form pro-inflammatory nanocrystalline complexes with dsRNA that are recognized by TLR3 differently than dsRNA alone. dsRNA complexes that activate IL-6 production in NHEK and those that do not are both able to enter cells and co-localize with TLR3. However, the crystallinity of these AMP-dsRNA complexes, specifically the geometric spacing between parallel dsRNA and the repeat number of ordered dsRNA, strongly influences the level of TLR3 activation. Crystalline complexes that present dsRNA at a spacing that matches with the steric size of TLR3 can recruit and engage multiple TLR3 receptors, driving receptor clustering and immune amplification, whereas crystalline complexes that exhibit poor steric matching do not. Reverse-transcription quantitative PCR of IL-6 during siRNA knockdown of TLR3 confirms that cytokine production is due to TLR3: High levels of IL-6 transcription are observed for sterically matched complexes without TLR3 knockdown, whereas such activity is abrogated with TLR3 knockdown.